To date, there is no specific treatment that is effective against COVID-19. Therefore, all researchers worldwide are jointly conducting studies to determine which treatment is the best for dealing with the disease. Recently, studies showed that using the malaria drug chloroquine could help block infection with the Coronavirus. However, in a report issued by the FDA (Food and Drug Administration), there are cases where some people experience serious heart rate problems due to the use of the drug.
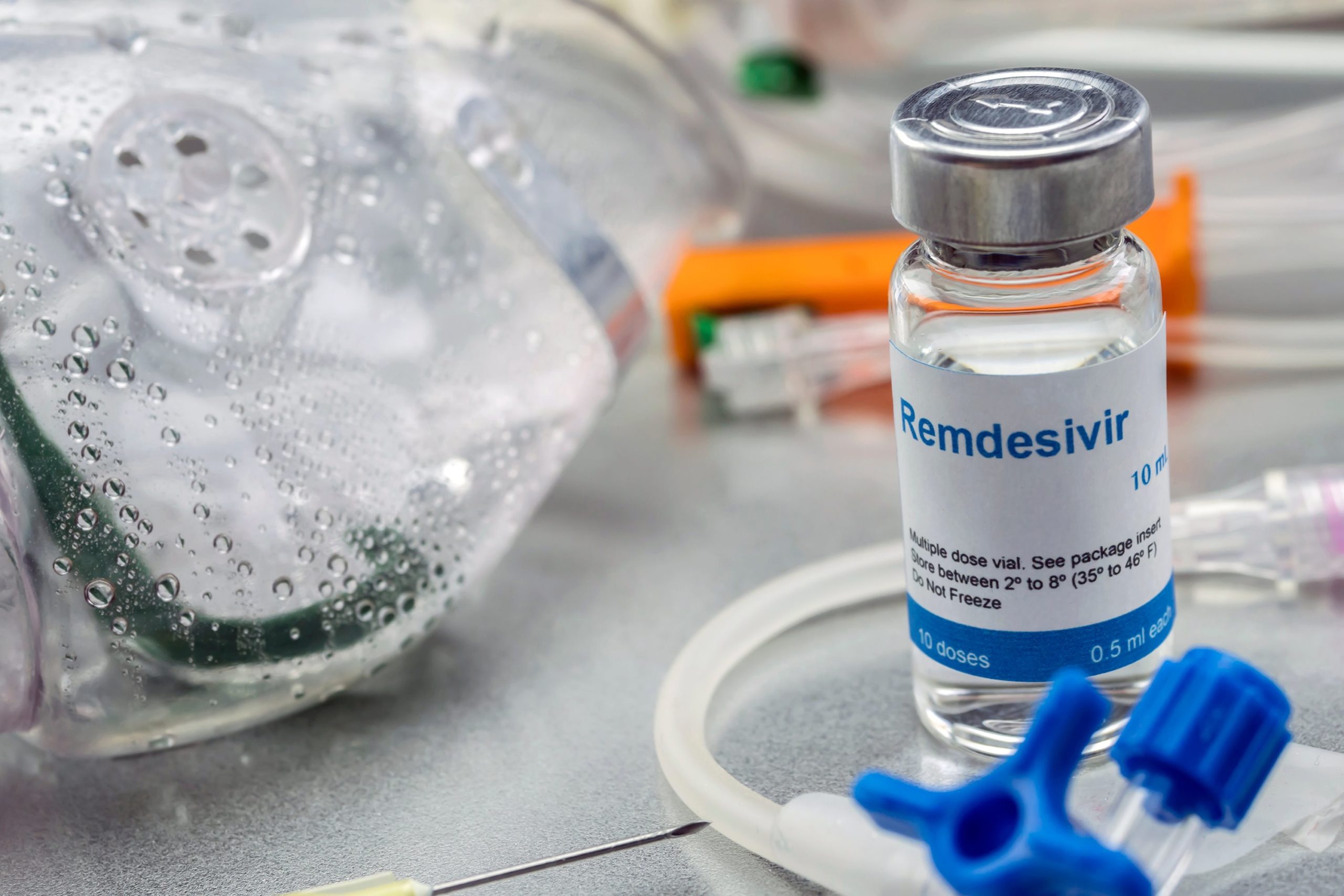
It does not stop there; recently, experts have often discussed the drug Remdesivir to help overcome COVID-19. However, is it Remdesivir? Remdesivir is an antiviral drug developed by the biotechnology company Gilead Sciences in the United States. Researchers have conducted trials of the drug Remdesivir in patients with COVID-19 with mild to moderate symptoms. As a result, this drug is known to speed up the recovery process of patients compared to those who were given a placebo drug.
So, how does the Remdesivir drug work? Coronaviruses use an enzyme called RNA-dependent RNA polymerase to copy their genetic material during the replication process inside infected cells. While Remdesivir is a nucleotide analogue, synthetic imitation of the molecules needed by the virus to replicate. Therefore, Remdesivir can help stop the replication process of the Coronavirus.
According to the results of a trial conducted by the National Institute of Allergy and Infectious Disease (NIAID), treatment using Remdesivir is known to speed up the recovery process of COVID-19 patients by up to 31% when compared to patients given placebo treatment. Patients on Remdesivir treatment required a median recovery time of 11 days, while on placebo treatment, 15 days. The mortality rate with Remdesivir treatment was also lower, at about 8%, compared to 11.6% for placebo treatment. The results of other studies also show that the drug Remdesivir has proven effective against several types of Coronaviruses in cell culture and is not toxic.
Text by Anggie Triana
Stock photos from: Google Search Images
Source(s):
- Medical News Today - FDA grant remdesivir emergency use for COVID-19 after turbulent week (2020). https://www.medicalnewstoday.com/articles/fda-grant-remdesivir-emergency-use-for-covid-19-after-turbulent-week, 10 May 2020.
- NIH NIAID - NIH Clinical Trial Shows Remdesivir Accelerates Recovery from Advanced COVID-19 (2020). https://www.niaid.nih.gov/news-events/nih-clinical-trial-shows-remdesivir-accelerates-recovery-advanced-covid-19, 10 May 2020.